- 缩略图
- 3D模型 68
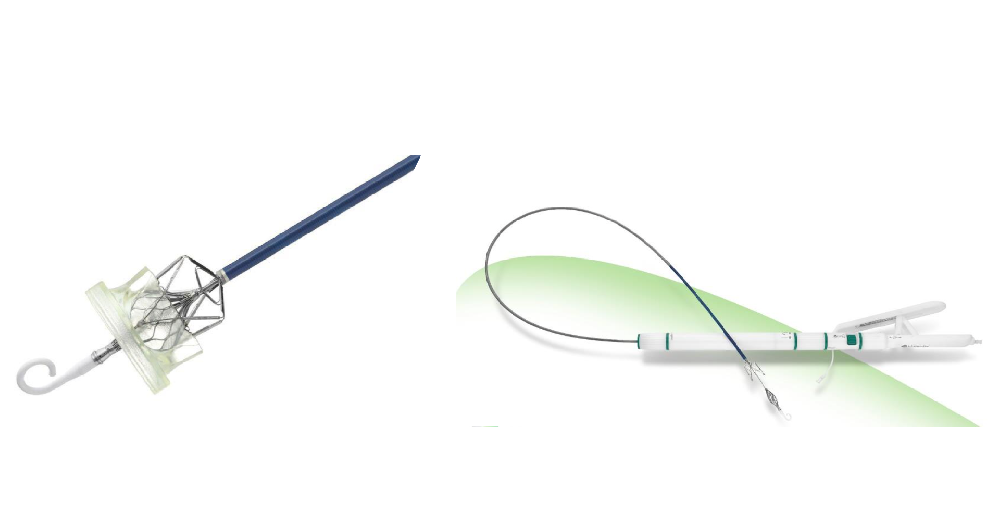
VenusA-Plus retains the advantage of strengthened radial force of VenusA-Valve, the first-generation valve replacement system of the Company, while thoroughly optimizing the delivery system and adding retrievable and repositionable features. This would effectively decrease the difficulty, promote successful rate, and improve postoperative benefits. VenusA-Plus will shorten the learning curve of doctors and help accelerate the development and popularization of TAVR in China.
VenusA-Plus was approved by NMPA on November 10th, 2020 and was commercially applied in the same year. On December 18th, 2020, VenusA-Plus was registered and approved by the Food and Drug Administration Committee of the Ministry of Public Health of Thailand.
其他产品