您现在的位置:首页 > 产品中心 > Cardiovalve
- 缩略图
- 3D模型 3
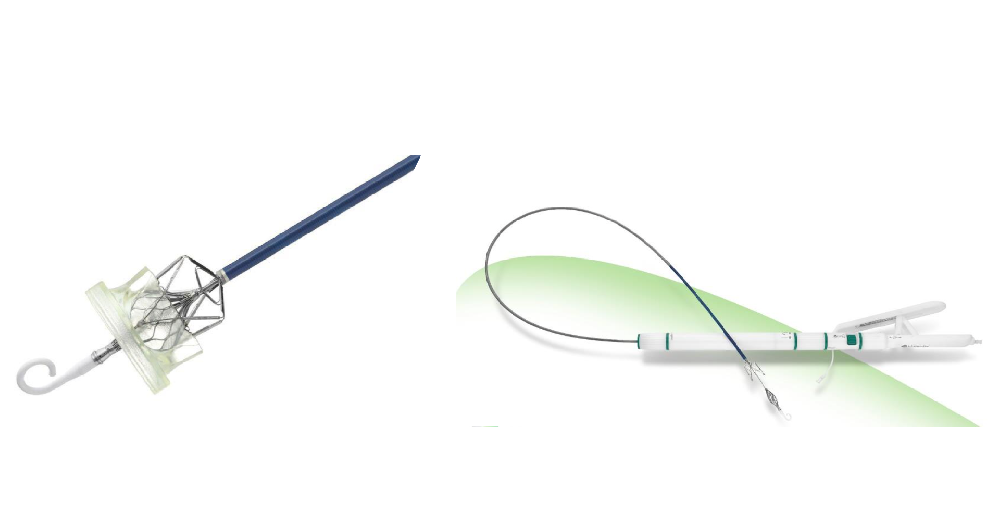
On December 7th, 2021, Venus Medtech announced the acquisition of Cardiovalve Ltd. The Cardiovalve system independently developed by the company is a transcatheter interventional replacement product for patients suffering from mitral or tricuspid regurgitation. Compared with products of the same kind, its transfemoral approach improves the safety of treatment in significant ways.
Cardiovalve’s treatment of mitral regurgitation has entered clinical trials in Europe and is currently in an early feasibility study in the U.S.. Furthermore, its device for the treatment of tricuspid regurgitation received 'Breakthrough Device Designation' by the FDA in January 2020 and entered an early feasibility study. It is worth mentioning that Cardiovalve is also the first privately held company to receive FDA's early feasibility study (EFS) approval for both TR and MR indications.
For more information, please click::http://www.cardiovalve.com/
其他产品