您现在的位置:首页 > 产品中心 > DragonFly™
- 缩略图
- 3D模型 0
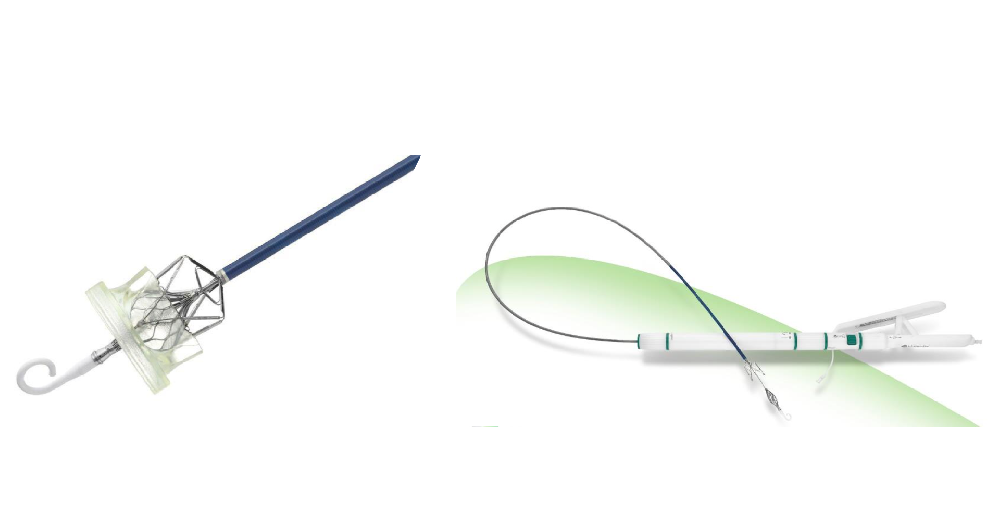
DragonFlyTM Transcatheter Mitral Valve Repair System (“DF”), independently developed by Valgen Medtech, is the first domestically engineered transfemoral mitral valve clip system approved in China. DragonFlyTM entered the NMPA's Special Review and Approval Procedures for Innovative Medical Devices in March 2021 and received marketing approval on November 29, 2023.
Venus Medtech and Valgen Medtech jointly announced that both parties reached an exclusive strategic marketing cooperation intention in relation to the DragonFlyTM Transcatheter Mitral Valve Repair System of Valgen Medtech on November 22nd, 2023. Both parties will carry out in-depth commercialization cooperation after the approval for listing of DragonFlyTM. Both parties will join hands and firmly believe that the cooperation will accelerate the development of the diagnosis and treatment of valvular heart diseases in China, and lay a solid foundation for building a comprehensive diagnosis and treatment platform for structural heart diseases in China in the future.
其他产品